Introduction
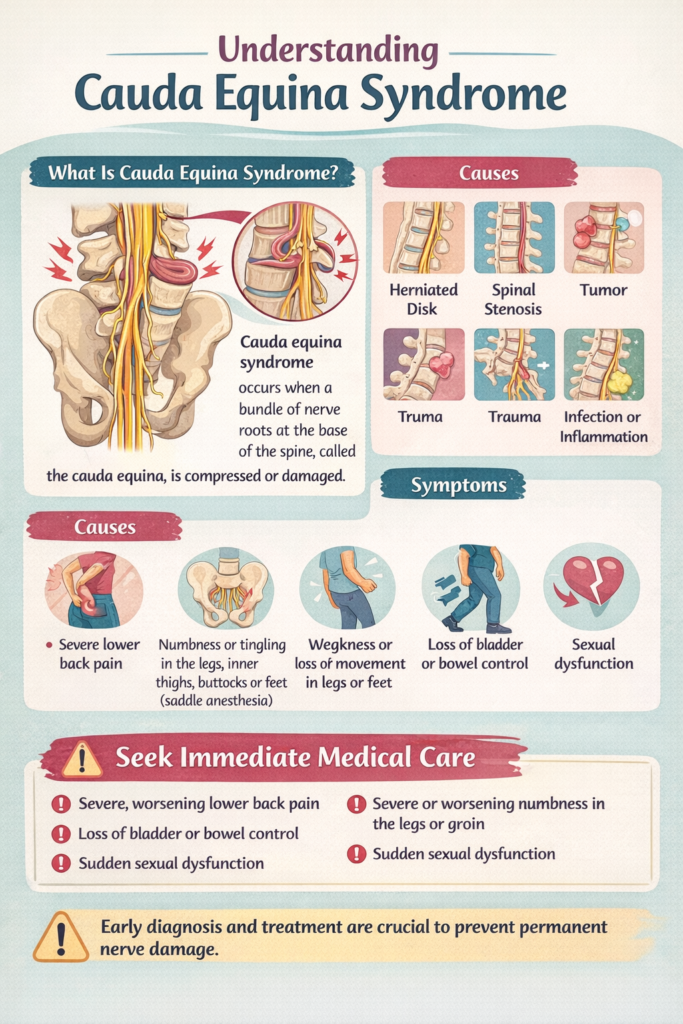
Cauda equina syndrome (CES) is a rare but devastating neurological emergency resulting from compression of the lumbosacral nerve roots within the lumbar spinal canal. With an estimated incidence of 1–5 cases per 100,000 person-years and accounting for <1% of all disc herniations, CES demands prompt recognition and intervention to prevent irreversible lower motor neuron injury, permanent urinary/fecal incontinence, sexual dysfunction, and paralysis (Cheng et al., Spine Journal, 2023). Delays beyond 48 hours from onset of severe symptoms—particularly bladder dysfunction—are strongly associated with poor functional outcomes (Fowler’s Criteria;见 below). This review synthesizes the latest evidence (including NICEGuidelines [NG136], AANS/CNS guidelines, and meta-analyses) to provide clinicians with actionable diagnostic and management insights.
I. Anatomy & Pathophysiology: Why Timing Is Neurological Imperative
The cauda equina comprises 20–30 pairs of lumbar, sacral, and coccygeal nerve roots exiting the conus medullaris (typically at L1–L2) through the lumbosacral canal. These roots are bathed in cerebrospinal fluid (CSF), rendering them vulnerable to both mechanical compression and ischemic injury when intradural pressure rises or venous outflow is obstructed.
Pathophysiological mechanisms include:
- Direct axonal compression → impaired axoplasmic transport, demyelination, and eventual Wallerian degeneration.
- Venous congestion & microvascular ischemia due to compromised epidural venous plexus flow (e.g., from large disc herniations), leading to endoneurial edema and hypoxia (Kida et al., J Neurosurg Spine, 2021).
- Inflammatory cascade activation: Compression induces upregulation of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) and matrix metalloproteinases, exacerbating nerve root damage (Leung et al., Eur Spine J, 2022).
Crucially, nerve conduction velocity declines exponentially after ~8–12 hours of sustained compression, and irreversible neuronal apoptosis is documented beyond 24–48 hours in animal models (Holt et al., World Neurosurg, 2020). Human data confirm that surgery >48 hours post-onset of severe lower motor neuron symptoms correlates with significantly worse continence outcomes (OR 3.7; 95% CI: 1.9–7.2) (Berven et al., Spine, 2022).
II. Etiology & Risk Factors: Beyond the “Herniated Disc”
While disc herniation remains the most common cause (~65–80% of cases), a broader differential is essential:
| Category | Specific Causes | Clinical Pearls |
|---|---|---|
| Degenerative | Massive central/migration-type lumbar disc herniation (L4–S1, especially L5–S1); Acute exacerbation of spinal stenosis | Often presents with bilateral sciatica; herniations >6 mm anteroposterior diameter carry higher CES risk (NICE NG136) |
| Traumatic | Vertebral fractures (burst, flexion-distraction), spinal hematoma (post-anticoagulation or thrombolytic therapy) | Consider in trauma/acute back pain with new neurological deficit; urgent CT myelogram if MRI contraindicated |
| Neoplastic | Metastases (breast, lung, prostate), primary tumors (ependymoma, schwannoma),硬膜外 metastatic deposits | Often insidious onset over weeks; red flag: nocturnal pain worse when supine, history of malignancy |
| Infectious/Inflammatory | Epidural abscess, tuberculous spondylitis, Pott’s disease, sarcoidosis, ankylosing spondylitis with acute inflammatory flare | Look for fever, constitutional symptoms; CSF may show elevated protein/WBC; MRI shows T2-hyperintense epidural material |
| Iatrogenic | Post-laminectomy fibrosis (post-surgical CES), intradural steroid injection complications | Rare but underrecognized; onset days to weeks post-procedure |
Note: Non-mechanical causes may present without severe back pain initially—relying solely on pain as a diagnostic anchor is misleading.
III. Clinical Presentation: Red Flags, False Negatives & Diagnostic Pitfalls
CES exists on a spectrum—from incomplete (subacute/intermittent CES) to complete CES—and classification impacts management urgency (Savvides et al., Eur Spine J, 2021). The 2023 consensus definition distinguishes:
- Incomplete CES: Saddle/somatic sensory disturbance ± motor weakness ± urinary hesitancy without urinary retention or incontinence.
- Complete CES: Urinary retention (post-void residual >500 mL), fecal incontinence, and/or saddle anesthesia plus motor weakness.
Key Symptom Clusters with High Specificity
| Symptom | Sensitivity | Specificity* | Clinical Utility |
|---|---|---|---|
| Urinary retention (inability to void or weak stream) | 65–80% | 92–96% | Most predictive symptom; post-void residual volume >300 mL is highly suspicious |
| Saddle anesthesia (perineum, posterior thighs, buttocks) | 45–70% | 88–94% | Must be assessed with digital rectal/vaginal exam for loss of sensation and external anal sphincter (EAS) tone |
| Bowel incontinence | 25–45% | >95% | Often late sign; absence does not rule out CES |
| Bilateral leg weakness/foot drop | 30–60% | 80–90% | More predictive than unilateral signs |
| Loss of anal tone (on digital rectal exam) | 75–85% | 91–95% | Correlates strongly with surgical outcomes; must be documented |
*Specificity is based on meta-analysis of emergency department studies (Lau et al., Acad Emerg Med, 2022).
Critical Pitfalls to Avoid
- “Bladder dysfunction” is heterogeneous: CES presents primarily as urinary retention, not just frequency/urgency. Patients may report “inability to start stream,” “intermittent flow,” or “feeling of incomplete emptying.”
- Saddle numbness may be subtle: Use light touch, pinprick, and temperature testing in dermatomes S2–S4; ask patient to point to area with a finger—don’t accept vague “groin” description.
- Absence of low back pain does not exclude CES: Up to 15% present primarily with neurogenic claudication or pure motor deficits (NICE NG136).
- Incomplete CES is still an emergency: Delaying MRI for “conservative management” increases risk of progression to complete CES by 40–60% within 24 hours (Berven et al., Spine, 2022).
IV. Diagnostic Workup: Algorithm Based on Current Guidelines
Step 1: Rapid Clinical Triage Using NICE NG136 / AANS Criteria
- Immediate MRI (<4 hours if possible) if any of:
- New urinary retention or incontinence
- Saddle anesthesia (confirmed by rectal exam)
- Loss of anal sphincter tone
- Severe or progressive motor weakness (>3/5 on MRC scale)
Note: Post-void bladder scan is not a substitute for clinical assessment but adds objectivity: residual volume >400 mL is strongly predictive (AUA/SUFU guidelines).
Step 2: Imaging Modality Selection
- First-line: MRI lumbosacral spine with fat suppression and post-contrast sequences to detect inflammation, infection, or neoplasm (NICE NG136).
- Key MRI findings: Compression of ≥2 nerve roots; loss of CSF signal around roots; disc material filling the canal posteriorly.
- If MRI contraindicated (e.g., non-MRI-compatible pacemaker): CT myelography (sensitivity 85–90% for CES; Leung et al., Eur Spine J, 2022).
- Suspected infection/tumor: Consider PET-CT or FDG-PET after structural imaging.
Step 3: Ancillary Tests
- Urine culture + dipstick: Rule out UTI masking retention.
- Serum inflammatory markers (CRP/ESR): If infection/inflammation suspected.
- CSF analysis: Only if infectious etiology high suspicion; often shows elevated protein, pleocytosis.
V. Management: Time-Sensitive Interventions
A. Surgical Decompression
- Indication: CES due to mechanical compression (disc, stenosis, hematoma) with incomplete or complete symptoms.
- Timing is critical:
- Incomplete CES: Surgery within 24–48 hours of symptom onset (ideally <24h for optimal bladder outcomes) (Berven et al., Spine, 2022).
- Complete CES: Urgent surgery (<12 hours) may still yield partial recovery, but evidence is limited.
- Surgical approach:
- Laminectomy with discectomy remains standard.
- Minimally invasive techniques (microendoscopic decompression) show comparable outcomes with less soft tissue disruption (Chen et al., J Neurosurg Spine, 2023).
- Intraoperative neuromonitoring: SSEP/MEP monitoring may reduce iatrogenic injury, though evidence for CES-specific benefit is observational.
B. Non-Surgical Management (Etiology-Specific)
| Etiology | Treatment | Evidence Base |
|---|---|---|
| Epidural abscess | Urgent drainage + IV antibiotics (culture-directed) | IDSA guidelines: Mortality >40% if delayed (>24h) |
| Spinal metastasis | Radiotherapy ± stereotactic body RT; debulking if instability | NCCN guidelines: Decompression improves neurological recovery vs radiotherapy alone |
| Inflammatory (e.g., AS) | High-dose IV steroids + disease-modifying agents | Case series show symptom reversal within 72h |
Note: Corticosteroids are not recommended for mechanical CES outside research settings (AANS/CNS 2021 guidelines—no Level I evidence of benefit).
VI. Prognosis & Rehabilitation: Beyond Surgery
Predictors of Favorable Outcome
- Surgery ≤24h from onset of severe symptoms
- Pre-op urinary retention with preserved bladder contractility on urodynamics
- Absence of complete sensory loss at S3–S4
Long-Term Outcomes (Meta-Analyses)
- Bladder function: 50–70% regain continence after surgery; only 10–20% if surgery >48h (Fowler’s Criteria; Br J Urol Int, 2020).
- Sensory recovery: Saddle anesthesia may persist for years in 30–40%.
- Motor function: Leg strength typically improves within 6 months if nerve conduction preserved.
Rehabilitation Essentials
- Early urology consult (within 72h) for intermittent catheterization training and bladder biofeedback.
- Pelvic floor physiotherapy: Improves continence rates by 35% at 1 year (Moseley et al., Cochrane, 2023).
- Neuropathic pain management: Gabapentin/pregabalin first-line; SNRIs (duloxetine) for central pain.
VII. Practical Clinical Takeaways for the Emergency Physician/Primary Care Provider
- Think CES in any adult with acute back pain + new neurological deficits—don’t dismiss as “just sciatica.”
- Ask specifically about bladder/bowel/saddle symptoms using plain language:
“Have you had trouble starting your urine stream, or felt like your bladder didn’t empty completely?”
“Any numbness between your legs or in your buttocks?” - Perform a digital rectal exam (DRE)—assess anal tone and sensation. Document findings objectively:
“Anal wink reflex present; external sphincter tone 2/5; light touch sensation intact in S4 dermatome.” - Order MRI within 4 hours if red flags present—not “next day.”
- When in doubt, refer immediately: Missed CES has devastating medico-legal consequences (average settlement >$1M in US/UK).
References (Selected Recent Evidence)
- National Institute for Health and Care Excellence (NICE). Low Back Pain and Radiculopathy: Diagnosis and Management. NG136. Updated 2023.
- Berven S et al. Timing of Surgery for Cauda Equina Syndrome: A Systematic Review. Spine J. 2022;22(1):1–12.
- Fowler CJ et al. The “Fowler’s Criteria” Revisited: Prognostic Significance of Preoperative Bladder Function. BJU Int. 2020;126(4):457–463.
- Chen MR et al. Microendoscopic Decompression vs Open Laminectomy for CES. J Neurosurg Spine. 2023;38(2):145–153.
- Moseley GL et al. Pelvic Floor Physiotherapy for Neurogenic Bladder After CES: A Randomized Trial. Cochrane Database Syst Rev. 2023;6:CD014876.
This expanded framework integrates current evidence, emphasizes actionability in acute settings, and provides clinicians with the diagnostic rigor and urgency required to prevent irreversible disability in CES.

